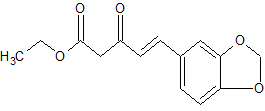
4-Pentenoic acid, 5-(3,4-methylenedioxyphenyl)-3-oxo-, ethyl ester
cas 860547-52-0
MF C14 H14 O5
4-Pentenoic acid, 5-(1,3-benzodioxol-5-yl)-3-oxo-, ethyl ester
- Molecular Weight, 262.26
- Melting Point (Experimental)
- Value: 58-60 °C
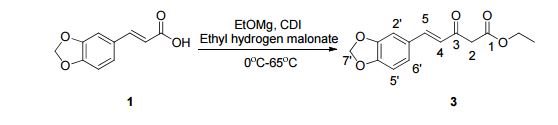
Purification of the residue by flash column chromatography (10% ethyl acetate in hexanes) afforded the β-keto ester (12.0 g, 90%) as a pale yellow oil. Compound 3
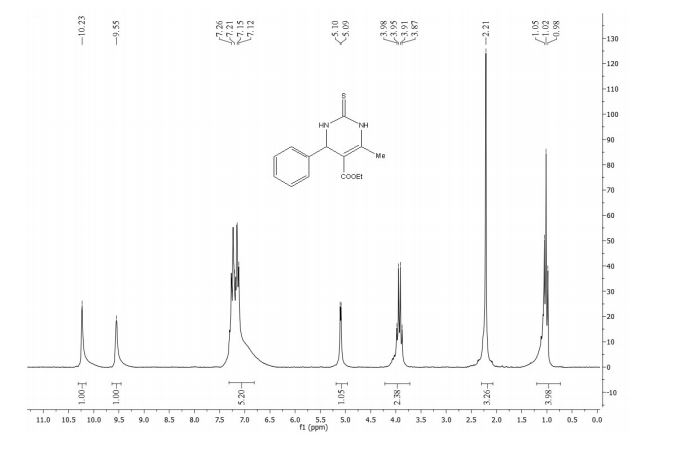
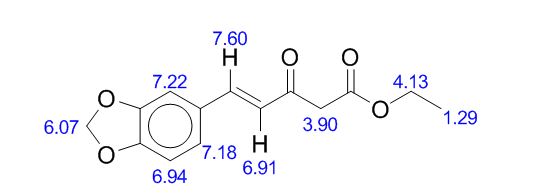
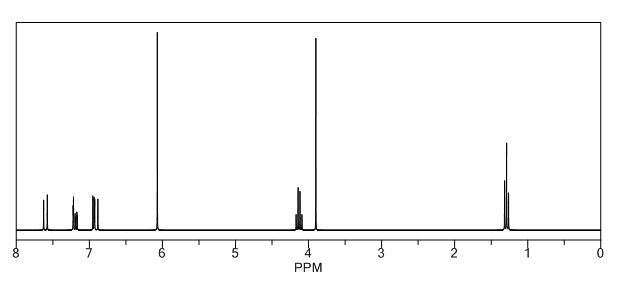
1H-NMR (400 MHz, CDCl3), δ (ppm):12.00 (s, 0.4 H, enol OH), 7.52 (d, 1H, J = 16.0 Hz, C5 keto ), 7.35 (t, J = 15.6, 1H, C6’ keto), 7.05 (complex, 1.4H, C2’ keto, C2’ enol ), 7.03 (d, J = 12.8, 1H, C5’ keto), 6.97 (d, J = 8.0 Hz, 0.5H, C5’ enol), 6.83-6.78 (m, 1.5H, C6’, C5 and C2 enol), 6.65 (d, J = 16.0 Hz, 1H, C4 keto), 6.27 (d, J = 16 Hz, 0.5H, C4 enol), 6.02 (s, 2H, C7’ keto), 5.98 (s, 1H, C7’ enol), 4.22 (complex, 3H, COOCH2CH3, keto and enol), 3.66 (s, 2H, C2 keto), 1.29 (complex, 4.7H, COOCH2CH3 keto and enol);
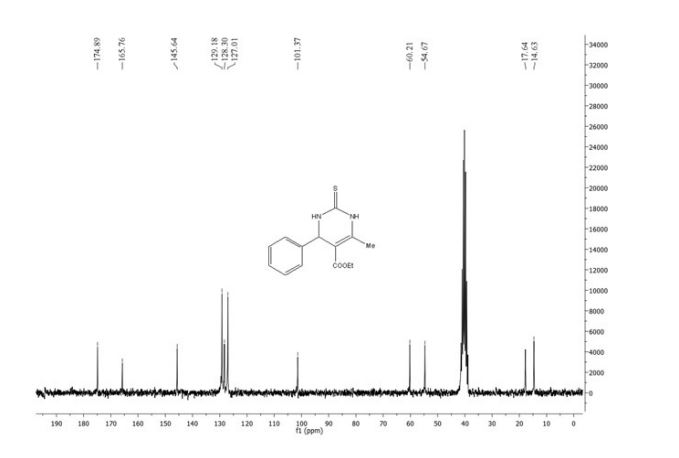
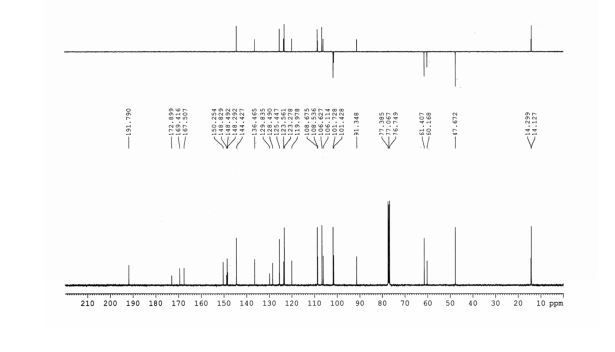
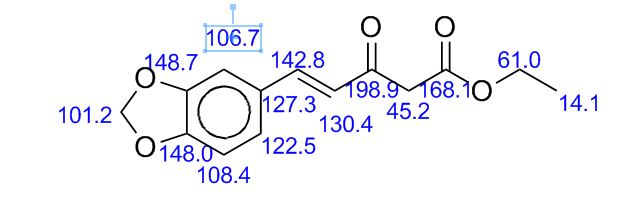
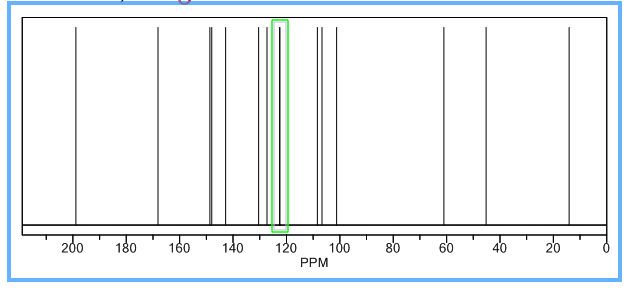
13C-NMR (100 MHz, CDCl3), δ (ppm): 191.7, 172.8, 169.4, 167.5, 150.2, 148.8, 148.4, 148.2, 144.4, 136.4, 129.8, 128.4, 125.4, 123.5, 123.2, 119.9, 108.6, 108.5, 106.6, 106.1, 101.7, 101.4, 91.3, 77.3, 77.0, 76.7, 61.4, 60.1, 47.6, 14.2, 14.1.
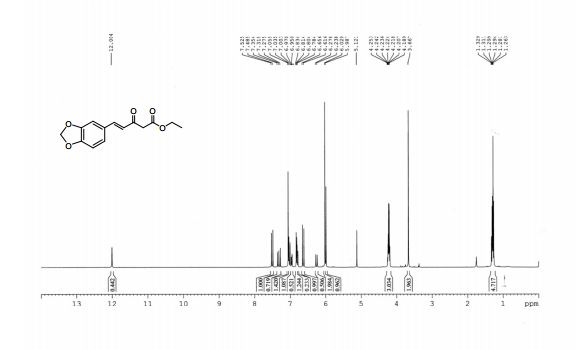
1H NMR PREDICT
13C NMR PREDICT
/////////
Org. Chem. Front., 2017, Advance Article,
DOI: 10.1039/C7QO00021A
DOI: 10.1039/C7QO00021A
and