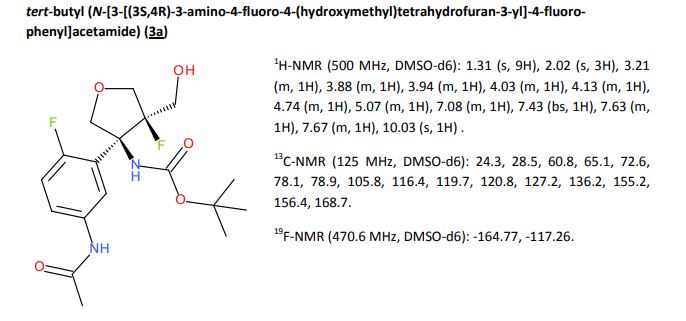
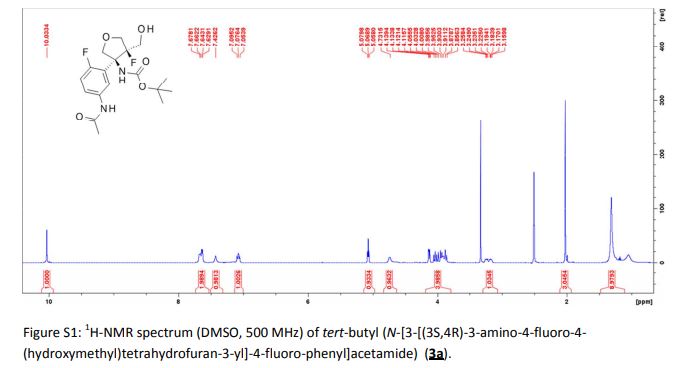
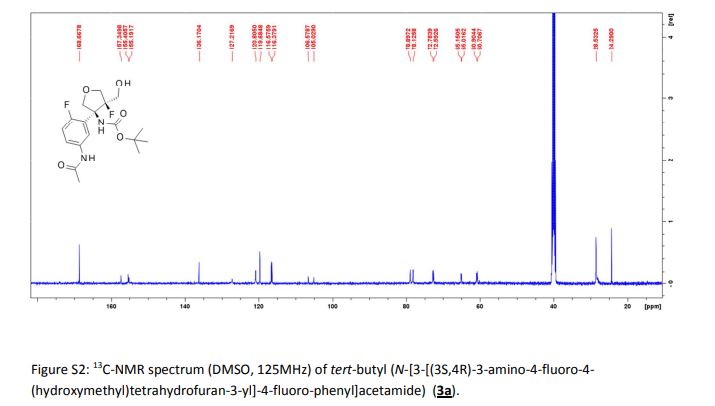
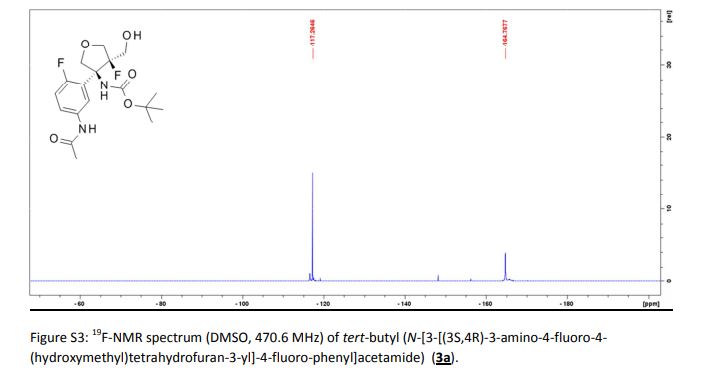
CH4630808
syn https://newdrugapprovals.org/2018/07/02/ch4630808/
Paper
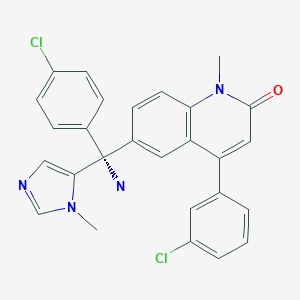
Development of a Kilogram-Scale Synthesis of a Novel Anti-HCV Agent, CH4930808
CH4630808 corrected
† Research Division, Chugai Pharmaceutical Co., Ltd., 1-135 Komakado, Gotemba, Shizuoka 412-8513, Japan
‡ Pharmaceutical Technology Division, Chugai Pharmaceutical Co., Ltd., 5-5-1 Ukima, Kita-ku, Tokyo 115-8543, Japan
§ Department of Chemistry for Materials, Graduate School of Engineering, Mie University, Tsu, Mie 514-8507, Japan
Org. Process Res. Dev., 2018, 22 (2), pp 236–240
DOI: 10.1021/acs.oprd.7b00383
Abstract

Herein, we report the kilogram-scale synthesis of CH4930808 (1) CH 4630808 CORRECTED, a novel anti-hepatitis C virus agent. While pursuing improved productivity using many through-process strategies, we conducted scrupulous impurity control. Finally, we successfully developed a practical and scalable process for the synthesis of (1·1.5Na·2.5H2O), by which we prepared 3.28 kg of the active pharmaceutical ingredient for clinical studies
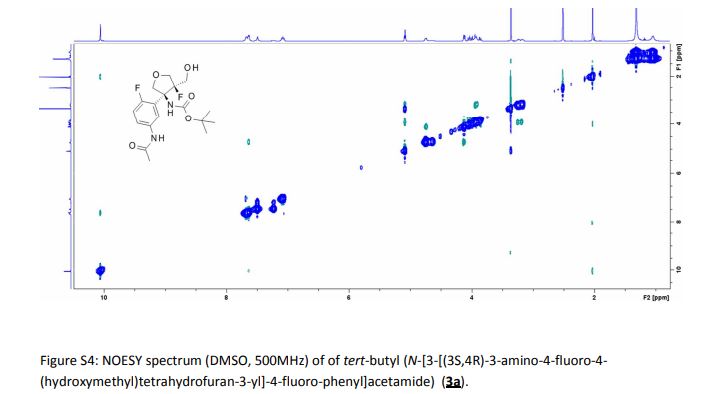
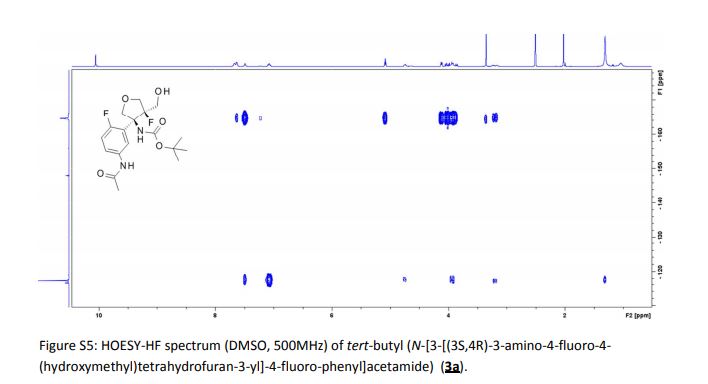
1H-NMR and 13C-NMR spectra of compound 5·HCl S 3– S 4


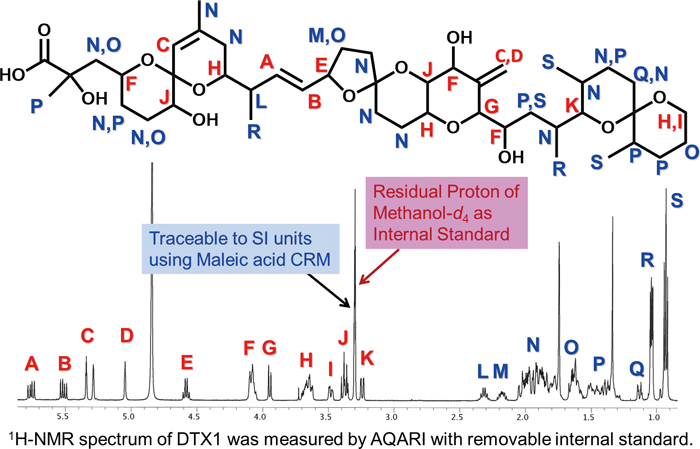
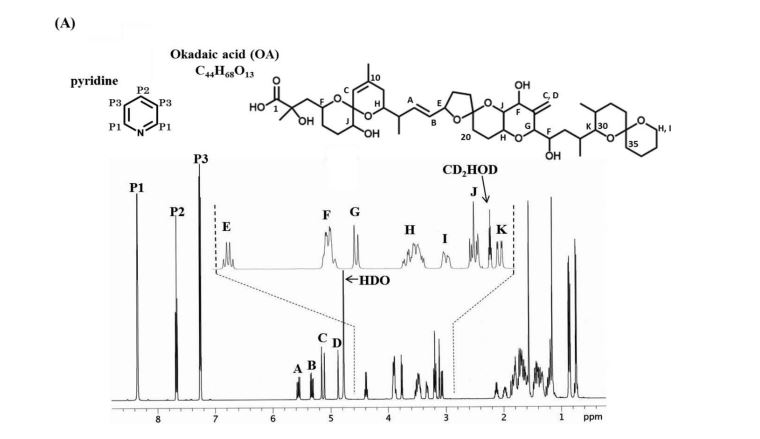
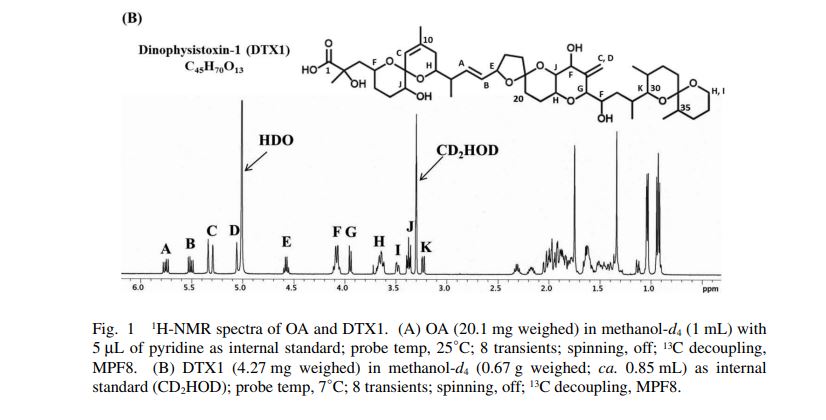
1H-NMR spectra of compound 1·1.5 Na·2.5 H2O S 5



13C-NMR spectra compound 1·1.5 Na·2.5 H2O S 6



1H-COSY spectra of compound 1·1.5 Na·2.5 H2O S 7 – S 8


DEPT spectra of compound 1·1.5 Na·2.5 H2O S 9 – S 10


HMBC spectra of compound 1·1.5 Na·2.5 H2O S 11 – S 17





MASS

////////////////