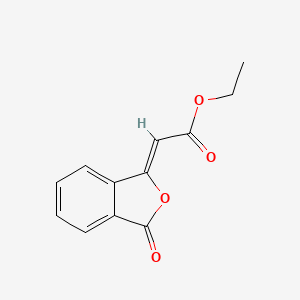
Ponalrestat
Phase III
An aldose reductase inhibitor potentially for the treatment of diabetes.
Imperial Chemical Industries Limited innovator
ICI-128436; MK-538; ICI-plc
CAS No.72702-95-5
Statil; Statyl;
3-[(4-Bromo-2-fluorophenyl)methyl]-3,4-dihydro-4-oxo-1-phthalazineacetic acid
Statil™ (3-(4-bromo-2-fluorobenzyl)-4-oxo-3H-phthalazin-1-ylacetic acid)
| Molecular Formula | C17H12BrFN2O3 |
| Molecular Weight | 391.19 |
IC50:Aldose reductase: IC50 = 7 nM (bovine); Aldose reductase: IC50 = 16 nM (rat); Aldose reductase: IC50 = 21 nM (pig); Aldose Reductase: IC50 = 21 nM (human); Rattus norvegicus:
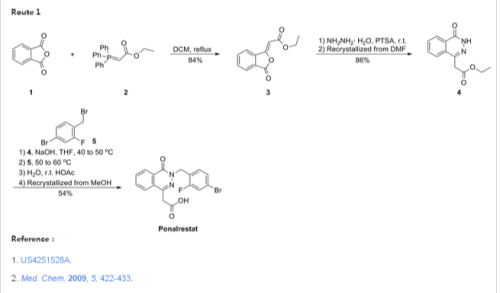
Medicinal Chemistry, 2009, Vol. 5, No. 5,
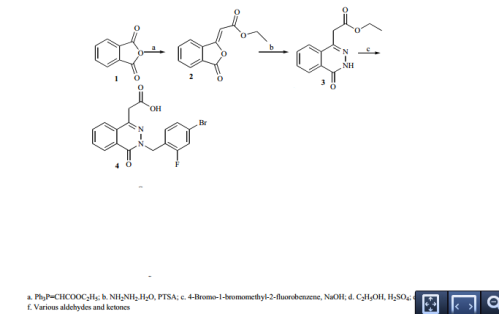
Synthesis of ethyl 2-(3-oxo-1,3-dihydro-1-isobenzofurany liden)acetate (2) A solution of phthalic anhydride (1.0 equiv.) and ethyl 2- (1,1,1-triphenyl-5 -phosphanylidene)acetate (1.1 equiv.) in 300 ml of dichloromethane (DCM) was refluxed for 3 hr. DCM was removed by vacuum at 40-50 o C. 2×150 ml of hexane was added to the resulting sticky solid, stirred for 10 min and the un-reacted 2-(1,1,1-triphenyl-5 -phosphanylidene)acetate was removed by filtration. The organic solvent was removed under vacuum and the resulting crude semisolid was taken to next step without further purification. Yield: 84%. 1 H-NMR CDCl3; (ppm): 1.1 (t, 3H), 4.2 (q, 2H), 6.0 (s, 1H), 7.6 (t, 1H), 7.7 (t, 1H), 7.8 (d, 1H), 8.9 (d, 1H). S
Synthesis of ethyl 2-(4-oxo-3,4-dihydro-1-phthalazinyl) acetate (3) A mixture of 2 (1.0 equiv.), hydrazine hydrate (0.8 equiv) and PTSA (1.0 equiv.) was ground by pestle and mortar at room temperature for 8 min. On completion, as indicated by TLC, the reaction mixture was treated with water. The resultant product was filtered, washed with water and recrystallized from DMF to give 3 in high yields (86%).1 H-NMR CDCl3; (ppm): 1.1 (t, 3H), 3.9 ( s, 2H), 4.1 (q, 2H), 7.6
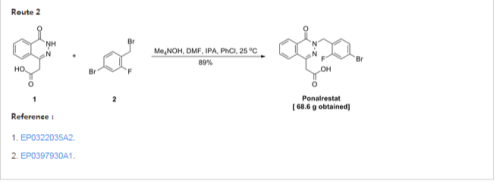
Synthesis of 2-[3-(4-bromo-2-fluorobenzyl)-4-oxo-3,4- dihydro-1-phthalazinyl]acetic acid (4)
A mixture of 3 (1.0 equiv.), NaOH (5.0 equiv.), and THF was stirred for 30 min at 40-50 o C. 4-bromo-1-bromomethyl-2-fluoro benzene (1.1 equiv.) was added to the reaction mixture and stirred for 2 hr at 50-60 o C. Water was added to the reaction mixture and stirred at room temperature for 1 hr. pH was adjusted to 2-3 using cold acetic acid. THF was removed and the aqueous phase was extracted with ethyl acetate (2×50 ml), washed with brine, dried over sodium sulphate and evaporated. The solid was crystallized with methanol to give 4 with 54 % yield.
1H-NMR (DMSOd6); (ppm): 3.98 (s, 2H), 5.3 (s, 2H), 7.17 (t, 1H), 7.35 ( dd, 1H, J1= 8.0, J2= 1.6), 7.55 (dd, 1H, J1= 8.0, J2= 1.6), 7.87 (t, 1H), 7.9 (t, 1H), 7.95 (t, 1H0, 8.29 (d, 1H).
///////////Ponalrestat, ICI-128436, MK-538, ICI-plc,
C1=CC=C2C(=C1)C(=NN(C2=O)CC3=C(C=C(C=C3)Br)F)CC(=O)O
take a tour
















take a tour




Cairo, Giza, Egypt










//////Cairo, Giza, Egypt,