CLICK……….
Cyclooxygenase (COX) or prostaglandin endoperoxide synthase (PGHS), catalyzes the conversion of arachidonic acid to inflammatory mediators such as prostaglandins (PGs), prostacyclins and thromboxanes. COX exists in mainly two isoforms: COX-1 and COX-2.Nonsteroidal anti-inflammatory drugs (NSAIDs), widely used for relief of fever, pain and inflammation, act by inhibiting COX catalyzed biosynthesis of inflammatory mediators.
However, the therapeutic use of classical NSAIDs is associated with well-known side effects at the gastrointestinal level (mucosal damage, bleeding) and, less frequently, at the renal level.
Two decades after the discovery of COX isoforms, it was recognized that selective inhibition of COX-2 might be endowed with improved anti-inflammatory properties and reduced gastrointestinal toxicity profiles than classical NSAIDs.
Overall, these selective COX-2 inhibitors (coxibs) have fulfilled the hope of possessing reduced risk in gastrointestinal events, but unfortunately cardiovascular concerns regarding the use of these agents have emerged that led to the withdrawal of rofecoxib (Vioxx) and valdecoxib (Bextra) from the market in 2004 and 2005, respectively.
Ongoing safety concerns pertaining to the use of non-selective NSAIDs have spurred development of coxibs with improved safety profile.
……………………………………………………………………………………………..

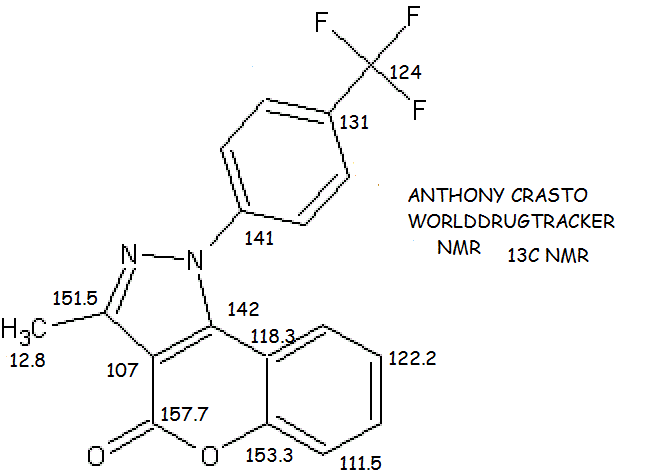
cas no 1616882-93-9
- mf……….C18 H11 F3 N2 O2
- [1]Benzopyrano[4,3-c]pyrazol-4(1H)-one, 3-methyl-1-[4-(trifluoromethyl)phenyl]-
3-Methyl-1-(4-(trifluoromethyl)phenylchromeno[4,3-c]pyrazol-4(1H)-one
Scheme 1.
Reagent and conditions: (a) Piperidine, rt, 20 min; (b) ArNHNH2, EtOH, reflux, 5 h; (c) K2CO3, acetone, reflux, 24 h.
COMPD IS
- 3-Methyl-1-(4-(trifluoromethyl)phenylchromeno[4,3-c]pyrazol-4(1H)-one (3b):
- White solid; yield 90%; mp: 224–225 °C;
- 1H NMR (CDCl3, 400 MHz): δ ppm 7.89 (d, 2H, J = 8.32 Hz, Ar-H), 7.73 (d, 2H, J = 8.24 Hz, Ar-H), 7.45–7.52 (m, 2H, H-6, H-7), 7.16 (dd, 1H, J = 1.4, 8.2 Hz, H-9), 7.10 (td, 1H, J = 1.56, 7.38 Hz, H-8), 2.69 (s, 3H, CH3);
- 13C NMR (CDCl3, 100 MHz): δ ppm 157.7, 153.3, 151.5, 142.3, 141.8, 131.9, 127.2, 127.1, 127.0, 124.0, 122.2, 118.3, 111.5, 107.1, 12.8;
- HRMS (ESI) m/z: Calcd for C18H11F3N2O2Na [M + Na]+ 367.0670; found 367.0676.
Synthetic Communications (2014), 44(13), 1914-1923
Unprecedented cyclization was observed during N-sulfonylation of 3-[1-(phenylhydrazono)-ethyl]-chromen-2-one in pyridine, affording 3-methyl-1-phenylchromeno[4,3-c]pyrazol-4(1H)-ones. To avoid use of noxious pyridine, reaction was tried in different basic conditions and the best results were obtained with potassium carbonate in acetone. A wide range of substrates bearing either electron-donating or electron-withdrawing substituents on phenylhydrazine ring were compatible with the developed methodology. Rapid access of starting material, 3-acetylcoumarin, excellent yields of products, and use of environmentally benign base and solvent for the cyclization make this strategy an efficient and convenient method for synthesis of 3-methyl-1-phenylchromeno[4,3-c]pyrazol-4(1H)-ones.
Methyl-1-(4-(trifluoromethyl)phenylchromeno[4,3-c]pyrazol-4(1H)-one (4b):
Whitesolid;
yield 90%; mp: 224–225 °C;
1H NMR (CDCl3, 400 MHz):δppm
2.69 (s, 3H, CH3),
7.10(td, 1H,J= 1.56, 7.38 Hz, H-8),
7.16 (dd, 1H,J= 1.4, 8.2 Hz, H-9),
7.45–7.52 (m, 2H, H-6, H-7),
7.73 (d, 2H,J= 8.24 Hz, Ar-H),
7.89 (d, 2H,J= 8.32 Hz, Ar-H);
13C NMR (CDCl3, 100MHz):
δppm
12.8,
107.1,
111.5,
118.3,
122.2,
124.0,
127.0,
127.1,
127.2,
131.9,
141.8,
142.3,
HRMS (ESI)m/z: Calcd for C18H11F3N2O2Na [M + Na]+367.0670; found367.0676.
3-Methyl-1-(4-(trifluoromethyl)phenylchromeno[4,3-c]pyrazol-4(1H)-one
SEE BELOW 1H NMR, 13CNMR, AND MASS SPEC

13C NMR

MASS
References
1. Jones, G.; Willett, P.; Glen, R. C.; Leach, A. R.; Taylor, R. J. Mol. Biol. 1997, 267, 727.
2. Bernstein, F. C.; Koetzle, T. F.; Williams, G. J. B.; Meyer, E. F.; Brice, M. D.; Rodgers, J. R.; Kennard, O.; Shimanouchi, T.; Tasumi, M. J. Mol. Biol. 1977, 112, 535.




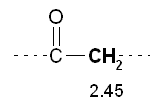 5) The peak 2 at d 2.09 has multiplicity 1 then refers to hydrogens that do not have near no hydrogen (m - 1 0 = H). This confirms the previous inference that the peak is due to a CH 3 near the bunker.
5) The peak 2 at d 2.09 has multiplicity 1 then refers to hydrogens that do not have near no hydrogen (m - 1 0 = H). This confirms the previous inference that the peak is due to a CH 3 near the bunker.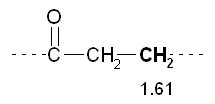 The peak 1 at d 2.45 has multiplicity 3 then refers to hydrogens which have close idogeni 2 (m - 1 = 2 H). This confirms the previous inference that the peak is attributable to a CH 2 near a bunker, and also suggests that there is a further CH 2 tied immediately over.
The peak 1 at d 2.45 has multiplicity 3 then refers to hydrogens which have close idogeni 2 (m - 1 = 2 H). This confirms the previous inference that the peak is attributable to a CH 2 near a bunker, and also suggests that there is a further CH 2 tied immediately over.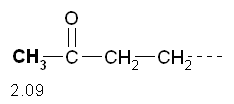 The peak 3 at d 1.61 has multiplicity 6 then refers to hydrogens that are near 5 hydrogens (m - 1 = 5 H). This confirms the previous inference that the peak is due to a CH 2 secondary that has near the two hydrogens of the peak 1 and the other three hydrogen atoms, those of the CH 3terminal.
The peak 3 at d 1.61 has multiplicity 6 then refers to hydrogens that are near 5 hydrogens (m - 1 = 5 H). This confirms the previous inference that the peak is due to a CH 2 secondary that has near the two hydrogens of the peak 1 and the other three hydrogen atoms, those of the CH 3terminal.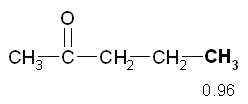 The peak 4 at d 0.96 has multiplicity 3 then refers to hydrogens which have close idogeni 2 (m - 1 = 2 H). This confirms the previous inference that the peak is due to the CH 3 terminal of the molecule.
The peak 4 at d 0.96 has multiplicity 3 then refers to hydrogens which have close idogeni 2 (m - 1 = 2 H). This confirms the previous inference that the peak is due to the CH 3 terminal of the molecule.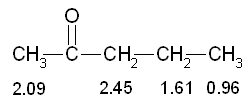 6) The deductions made so far lead us to hypothesize that the NMR spectrum is related to the molecule 2-pentanone which has the structure shown at right:
6) The deductions made so far lead us to hypothesize that the NMR spectrum is related to the molecule 2-pentanone which has the structure shown at right: