Therapeutic options for brain infections caused by pathogens with a reduced sensitivity to drugs are limited. Recent reports on the potential use of linezolid in treating brain infections prompted us to design novel compounds around this scaffold. Herein, we describe the design and synthesis of various oxazolidinone antibiotics with the incorporation of silicon.
Our findings in preclinical species suggest that silicon incorporation is highly useful in improving brain exposures. Interestingly, three compounds from this series demonstrated up to a 30-fold higher brain/plasma ratio when compared to linezolid thereby indicating their therapeutic potential in brain associated disorders
Design, Synthesis, and Identification of Silicon Incorporated Oxazolidinone Antibiotics with Improved Brain Exposure
† CSIR-National Chemical Laboratory, Dr. Homi Bhabha Road, Pune 411008,India
‡ Daiichi Sankyo India Pharma Pvt. Ltd., Gurgaon, Haryana 122015, India
§ Incozen Therapeutics Pvt. Ltd., Alexandria Knowledge Park, Turkapally, Rangareddy 500078, India
ACS Med. Chem. Lett., Article ASAP
DOI: 10.1021/acsmedchemlett.5b00213
Publication Date (Web): October 26, 2015
Copyright © 2015 American Chemical Society
*E-mail: ds.reddy@ncl.res.in.
SILINEZOLID, NDS 10024
CAS 1430321-45-1
C18 H26 F N3 O3 Si, 379.50
Acetamide, N-[[(5S)-3-[4-(4,4-dimethyl-1-aza-4-silacyclohex-1-yl)-3-fluorophenyl]-2-oxo-5-oxazolidinyl]methyl]-
Examples from patent
- (S)—N((3-(4-(4,4-dimethyl-1,4-azasilinan-1-yl)-3-fluorophenyl)-2 oxooxazolidin-5-yl)methyl)acetamide
- NDS 10024
Patent US20140296133
SEE
AUTHORS
- Preparation of (S)—N((3-(4-(4,4-dimethyl-1,4-azasilinan-1-yl)-3-fluorophenyl)-2 oxooxazolidin-5-yl)methyl)acetamide (12)
- To a solution of 8 (50 mg, 0.135 mmol) in dimethylformamide (DMF), lithium-t-butoxide (LiOtBu) (32.3 mg, 0.4 mmol) is added. The mixture is stirred at 25° C. for 15 min, followed by the addition of MeOH (0.01 mL, 0.27 mmol). 6 (52 mg, 0.27 mmol) is then added and the reaction mixture is allowed to stir at 25° C. for 24 h. Glacial acetic acid is then added and the organic phase is extracted with EtOAc and washed with brine solution. The crude material is purified by column chromatography on silica gel using hexane-EtOAC mixtures to furnish the pure product 12. The analogous procedure for the corresponding morpholine analogue was adapted from Lu, C. V.; Chen, J. J.; Perrault, W. R.; Conway, B. G.; Maloney, M. T.; Wang, Y. Org. Pro. Res. and Development. 2006, 10, 272-277.
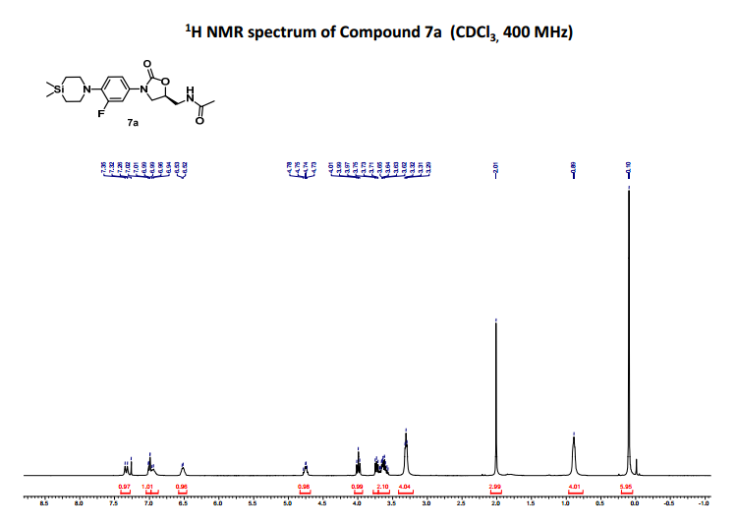
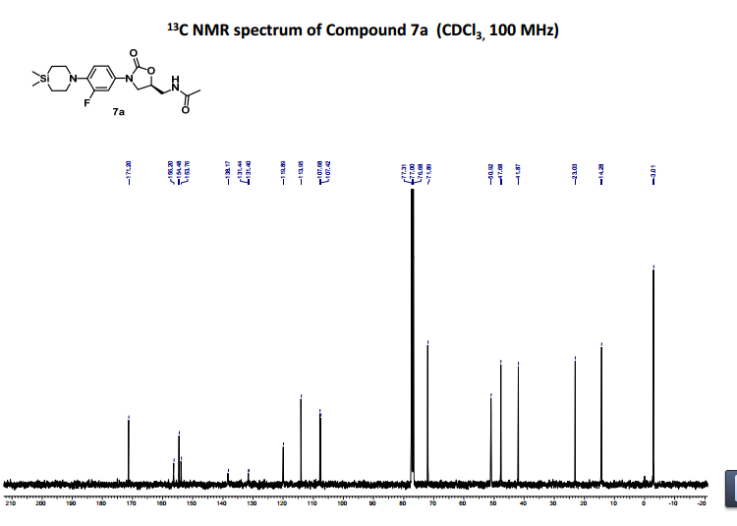
- 1H NMR (200 MHz, CDCl3): δ 7.33 (d, J=13.8 Hz, 1H), 7.02-6.94 (m, 2H), 6.52 (t, J=5.8 Hz, 1H), 4.77-4.73 (m, 1H), 3.99 (t, J=9.04 Hz, 1H), 3.72 (dd, J=9.0 Hz, 6.8 Hz, 1H), 3.69-3.58 (m, 2H), 3.31 (t, J=5.5 Hz, 4H), 2.01 (s, 3H), 0.89 (t, J=5.5 Hz, 4H), 0.10 (s, 6H). 13C NMR (100 MHz, CDCl3): δ171.2, 155.0 (d, J=244.3 Hz), 154.5, 138.2 (d, J=9.3 Hz), 131.5, 119.9, 114.0 (d, J=3.4 Hz), 107.6 (d, J=27.1 Hz), 71.9, 50.9, 47.7, 41.9, 23.0, 14.3, −2.9.
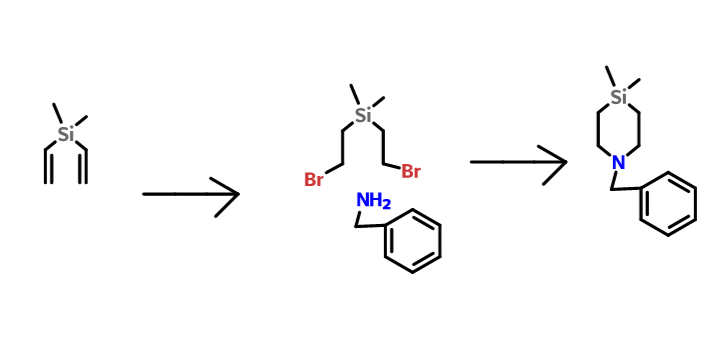
- Preparation of Bis(bromomethyl)dimethylsilane (2) (as per scheme 2)
- HBr gas is bubbled to a solution of dimethyl divinylsilane 1 (10.0 g, 89.28 mmols), and dibenzoylperoxide (DBP, 100 mg), in heptane (100 mL) at 0° C. for 30 min. The Reaction mixture (RM) is allowed to stir at room temperature (25° C.) for 18 h, water (200 mL) is added to the reaction mixture and the organic layer is separated. The heptane layer is washed with 2N NaOH (2 100 mL), dried and concentrated to obtain the product 2 as a colourless liquid (24.5 g) in 100% yield.
- 1H NMR (200 MHz, CDCl3): δ 3.58-3.49 (m, 4H), 1.45-1.40 (m, 4H), 0.09 (s, 6H).
- Benzylamine (20 mL, 182 mmol) and Et3N (15.2 mL, 109 mmol) are added to a solution of bis-(bromomethyl) dimethylsilane 2 (10 g, 36.5 mmol) in chloroform (100 mL). The mixture is then refluxed for 16 h. 5% sodiumhydroxide solution (150 mL) is then added and the aqueous layer is extracted with dichloromethane (DCM, 2×100 mL). It is then washed with brine (200 mL), dried and concentrated. The product is purified by column chromatography on silica gel using hexane-EtOAc mixtures to obtain the product 3 as a light yellow liquid (4.3 g) in 54% yield.
- 1H NMR (200 MHz, CDCl3): δ 7.23-7.35 (m, 5H), 3.66 (s, 2H), 2.68 (t, J=6.3 Hz, 4H), 0.75 (t, J=6.3 Hz, 4H), 0.04 (s, 6H).
- Preparation of 1-benzyl-4,4-dimethyl-1,4-azasilinane (3)
Preparation of 4,4-dimethyl-1,4-azasilinane hydrochloride (4)
- To a solution of 4,4-dimethyl-1,4-azasilinane 3 (2.3 g, 10.5 mmol) in EtOH (20 mL), 6N hydrochloricacid (1.75 mL, 10.5 mmol) is added and the solvent is removed under reduced pressure. The reaction mixture is co-evaporated with EtOH (2×10 mL) and recrystallized from EtOH-diethyl ether. To a slurry of Pd/C (50 mg) in EtOH (15 mL) an ethanolic solution of above prepared HCl salt is added drop wise and stirred at 25° C. under hydrogen atmosphere for 20 h. The reaction mixture is filtered through celite and washed with 2×20 mL of MeOH. The filtrate is then concentrated under reduced pressure to give viscous oil which was triturated with diethyl ether to obtain the product 4 as a white solid (950 mg) in 70% yield.
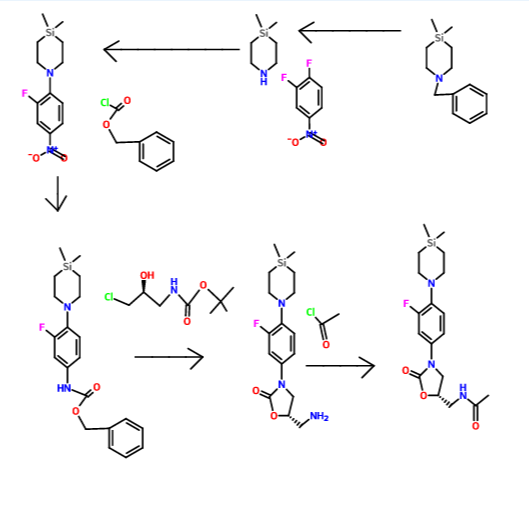
Preparation of 1-(2-fluoro-4-nitrophenyl)-4,4-dimethyl-1,4-azasilinane (9)
- To a solution of 4,4-dimethyl-1,4-azasilinane hydrochloride 4 (500 mg, 3.85 mmol) in EtOAc (15 mL), triethylamine (1.3 mL, 9.63 mmol) is added and stirred at 25° C. for 10 min. The reaction mixture is cooled to 0° C. and 3,4-difluoronitrobenzene (612 mg, 3.85 mmol) is added drop wise and allowed to stir at 25° C. for 6 h. Water is then added and the organic layer is separated. The aqueous layer is extracted with EtOAc (2×10 mL) and the solvent is removed under reduced pressure. The product is purified by column chromatography using hexane-EtOAc mixtures and a crystalline yellow solid 9 (721 mg) is obtained in 70% yield.
- 1H NMR (200 MHz, CDCl3): δ 7.93-7.84 (m, 2H), 6.86 (t, J=4 Hz, 1H), 3.70-3.67 (m, 4H), 0.91-0.85 (m, 4H), 0.12 (s, 6H). 13C NMR (50 MHz, CDCl3): δ 151.1 (d, J=246.71 Hz), 144.4 (d, J=7.13 Hz), 137.8 (d, J=8.59 Hz), 121.4, 115.9 (d, J=4.61 Hz), 113.2 (J=27.78 Hz), 49.4, 13.8, −2.8. IR (CHCl3): ν 2948, 2894, 1603, 1523, 1492, 1400, 1342, 1223, 983, 832, 742 cm−1′. M.P: 70-72° C.
Preparation of benzyl 4-(4,4-dimethyl-1,4-azasilinan-1-yl)-3-fluorophenylcarbamate (10)
- To a solution of compound 9 (610 mg, 2.28 mmol) in THF (25 mL), Pd/C (30 mg) is added and hydrogenated under a pressure of 35 psi in a par hydrogenator for 8 h. The reaction mixture is filtered through celite. Celite pad is washed with THF (2×20 mL). To the filtrate, saturated NaHCO3 (420 mg, 5.01 mmol) and CBzCl (427 mg, 2.5 mmol) are added at 0° C. and stirred at 25° C. for 5 h. 10 mL water is added to reaction mixture and the aqueous layer is extracted with EtOAc (2×20 mL). The crude mixture is then subjected to column chromatography on silica gel using hexane-EtOAc mixtures to afford the product as a viscous liquid 10 (690 mg) in 82% yield.
- 1H NMR (200 MHz, CDCl3): δ 7.41-7.37 (m, 5H), 6.94-6.93 (m, 2H), 6.68 (s, 1H), 5.21 (s, 1H), 3.3 (t, J=6.38 Hz, 4H), 0.93 (t, J=6.08 Hz, 4H), −0.13 (s, 6H). 13C NMR (50 MHz, CDCl3): 155.4 (d, 244.4 Hz), 153.6, 136.1, 135.9, 128.6, 128.5, 128.3, 120.4, 117.2 (d, 18.7 Hz), 114.7, 108.3 (20.5 Hz), 67.1, 51.4, 14.4, −3.0. IR (CHCl3): ν 3317, 2953, 2803, 1706, 1594, 1521, 1271, 1221, 1058, 869, 759 cm−1. M.P: 80-82° C.
Preparation of (S)-5-(aminomethyl)-3-(4-(4,4-dimethyl-1,4-azasilinan-1-yl)-3-fluorophenyl)oxazolidin-2-one (11) (NDS-10057)
- To a solution of 10 (1.20 g, 3.23 mmol) and (S)-tert-butyl 3-chloro-2-hydroxypropylcarbamate (1.35 g, 6.47 mmol) in DMF (10 mL), LiOtBu (1.03 g, 12.94 mmol) is added at 0° C. The mixture is stirred at 25° C. for 45 h. The starting material 10 is not consumed completely. Saturated NH4Cl is then added; the organic phase is extracted with EtOAc (2×20 mL), washed with brine solution, dried and concentrated. The crude residue is dissolved in 20 mL of DCM-TFA mixture (8:2) and stirred at 25° C. for 3 h. RM is concentrated and dissolved in water (10 mL), the aqueous layer is washed with diethyl ether (2×50 mL), basified with saturated NaHCO3 and extracted with DCM (2×50 mL). The DCM layer is dried and concentrated. The crude is purified by column chromatography on silica gel using hexane-EtOAc mixtures to obtain the product as an off-white solid (500 mg) in 45% (based on recovery of starting material) over 2 steps.
- 1H NMR (400 MHz, CDCl3): δ 7.36 (dd, J=14.2 Hz, 2.3 Hz, 1H), 7.09 (dd, J=8.8 Hz, 1.7 Hz, 1H), 6.96 (t, J=9.5 Hz, 1H), 4.72-4.59 (m, 1H), 4.00 (t, J=8.3 Hz, 1H), 3.79 (dd, J=8.7 Hz, 6.8 Hz, 1H), 3.30 (t, J=6.2 Hz, 4H), 3.03 (dq, J=13.6 Hz, 4.2 Hz, 2H), 0.90 (t, J=6.2 Hz, 4H), 0.10 (s, 6H). 13C NMR (100 MHz, CDCl3): δ 155.1 (d, J=244.3 Hz), 154.7, 137.9 (d, J=9.0 Hz), 132.1 (d, J=10.3 Hz), 112.0 (d, J=4.3 Hz), 113.8 (d, J=3.2 Hz), 107.4 (d, J=26.9 Hz), 73.8, 51.0, 47.8, 45.01, 14.4, −2.9. IR (CHCl3): ν 3685, 3021, 2955, 2809, 2401, 1747, 1515, 1416, 1219, 1029, 991, 870, 771, 667 cm−1. M.P: 94-96° C. ESI-MS: 360.11 (M+Na).
Preparation of (S)—N-((3-(4-(4,4-dimethyl-1,4-azasilinan-1-yl)-3-fluorophenyl)-2-oxooxazolidin-5-yl)methy)acetamide (12) (NDS 10024)
- To solution of amine 11 (300 mg, 0.9 mmol) and DIPEA (0.3 mL, 1.78 mmol) in dry THF (4.0 mL), acetylchloride (0.08 mL, 1.07 mmol) is added at 0° C., and stirred at 25° C. for 3 h. Further, saturated NaHCO3 (5.0 mL) is added to the reaction mixture and extracted with EtOAc (2×5 mL). The organic layer is washed with brine, dried and concentrated. The product is purified by column chromatography on silica gel using hexane-EtOAc mixtures to obtain the product as an off-white solid (170 mg) in 50% yield.
- 1HNMR (400 MHz, CDCl3): δ 7.33 (d, J=13.8 Hz, 1H), 7.02-6.94 (m, 2H), 6.52 (t, J=5.8 Hz, 1H), 4.77-4.73 (m, 1H), 3.99 (t, J=9.04 Hz, 1H), 3.72 (dd, J=9.0 Hz, 6.8 Hz, 1H), 3.69-3.58 (m, 2H), 3.31 (t, J=5.5 Hz, 4H), 2.01 (s, 3H), 0.89 (t, J=5.5 Hz, 4H), 0.10 (s, 6H). 13C NMR (100 MHz, CDCl3): δ171.2, 155.0 (d, J=244.3 Hz), 154.5, 138.2 (d, J=9.3 Hz), 131.5, 119.9, 114.0 (d, J=3.4 Hz), 107.6 (d, J=27.1 Hz), 71.9, 50.9, 47.7, 41.9, 23.0, 14.3, −2.9. IR (CHCl3): ν 2401, 1759, 1675, 1519, 1216, 759, 669 cm−1M.P: 123-126° C. ESI-MS: 380.10 (M+H).
SCHEME 1
SCHEME2
SCHEME 3
SCHEME 4
Dr. D. Srinivasa Reddy of NCL winner Shanti Swarup Bhatnagar Award 2015
see
Dr. Srinivasa Reddy of CSIR-NCL bags the
prestigious Shanti Swarup Bhatnagar Prize
sept 2015…………..http://www.biospectrumindia.com/biospecindia/news/222486/
The award comprises a citation, a plaque, a cash prize of Rs 5 lakh
The Shanti Swarup Bhatnagar Prize for the year 2015 in chemical sciences has been awarded to Dr. D. Srinivasa Reddy of CSIR-National Chemical Laboratory (CSIR-NCL), Pune for his outstanding contributions to the area of total synthesis of natural products and medicinal chemistry.
This is a most prestigious award given to the scientists under 45 years of age and who have demonstrated exceptional potential in Science and Technology. The award derives its value from its rich legacy of those who won this award before and added enormous value to Indian Science.
Dr. Reddy will be bestowed with the award at a formal function, which shall be presided over by the honourable Prime Minister. The award, named after the founder director general of Council of Scientific & Industrial Research (CSIR), Dr. Shanti Swarup Bhatnagar, comprises a citation, a plaque, a cash prize of Rs 5 lakh.
Dr. Reddy’s research group current interests are in the field of total synthesis and drug discovery by applying medicinal chemistry. He has also been involved in the synthesis of the agrochemicals like small molecules for crop protection. The total synthesis of more than twenty natural products has been achieved in his lab including a sex pheromone that attracts the mealy bugs and has potential use in the crop protection. On the medicinal chemistry front significant progress has been made by his group using a new concept called “Silicon-switch approach” towards central nervous system drugs. Identification of New Chemical Entities for the potential treatment of diabetes and infectious diseases is being done in collaboration with industry partners.
His efforts are evidenced by 65 publications and 30 patents. He has recently received the NASI-Reliance industries platinum jubilee award-2015 for application oriented innovations and the CRSI bronze medal. In addition, he is also the recipient of Central Drug Research Institute award for excellence in the drug research in chemical sciences and scientist of the year award by the NCL Research Foundation in the year 2013. Dr. Reddy had worked with pharmaceutical companies for seven years before joining CSIR-NCL in 2010.
AN INTRODUCTION
Ph.D., University of Hyderabad, 2000 (Advisor: Professor Goverdhan Mehta).
Post-doctoral with Profs. Sergey A. Kozmin(University of Chicago, USA) and Prof.
Jeffrey Aubé (University of Kansas, USA)
Experienced in leading drug discovery programs (Dr. Reddy’s & TATA Advinus – 7
years of pharma experience)
Acquired skills in designing novel small molecules and lead optimization
Experienced in planning and execution of total synthesis of biologically active
molecules with moderate complexity
One of the molecules is currently in human clinical trials.
MYSELF WITH HIM
OTHER AUTHORS


////////
C[Si]1(C)CCN(CC1)c2ccc(cc2F)N3C[C@H](CNC(C)=O)OC3=O