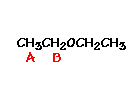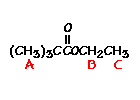Aromatic protons show up from 6.5-8.5 ppm. Benzylic protons are from 2–3 ppm.
Monosubstituted rings will have 5 protons in the region 6.5-8.5 ppm; disubstituted rings will have 4 protons; trisubstituted rings will have 3 protons (and so on). Examples of the NMR of aromatics of mono-, di-, and tri-substituted aromatics are shown below. When interpreting the spectrum of an aromatic compound, remember to count the number of protons in the aromatic region to determine how many times the ring is substituted.
If an aromatic ring has more than one substituent, careful analysis of the shifts and splitting pattern of the protons in the aromatic region reveals the positions on the ring of the different substituents. However, the the shifts depend on the substituents and the splitting is not really first order. Most sophomore-level courses do not cover NMR spectroscopy in the depth required to analyze the aromatic region. Therefore, you will not be able to designate the exact ring substition in many cases.
-substituted rings usually show two symmetric sets of peaks that look like doublets. The
-substitution NMR aromatic region pattern usually looks quite different than the patterns for both
substituted aromatic rings.
substitution are illustrated in the NMR spectra of different isomers of chloronitrobenzene, below. The CDCl
peak is pointed out in each spectrum. (The samples were run using CDCl
, which shows up at 7.24 ppm. This is used to calibrate the spectrum.) Note the symmetry of the
substituted chloronitro benzene. (Click on each full-size image to view details of the region from 6.5-8.5 ppm.)
Trimethyl 2-Hydroxy-2-(2-methoxy-2-oxoethyl)-4-(4-methylphenyl)-6-oxo-1,3,5-cyclohexanetricarboxylate
Edmont V. Stoyanov
Faculty of Pharmacy, Medical University of Sofia, Dunav 2, BG-1000 Sofia, Bulgaria
Tel.: (+359 2) 988 3142; Fax: (+359 2) 987 9874; E-mail: estoyanov@yahoo.com
Aromatic aldehydes react with dimethyl acetonedicarboxylate in molar ratio 1:2 with spontaneous intermolecular Michael addition to give polysubstituted cyclohexanones [1]. We report now the synthesis of an analogous product from 4-methylbenzaldehyde.
To a solution of 4-methylbenzaldehyde (1.20 g, 10 mmol) and dimethyl acetonedicarboxylate (3.48 g, 20 mmol) in 25 ml ethanol, 0.3 ml piperidine was added. The reaction mixture was left to stay at room temperature for 3 days. The separated crystals were filtered off, washed with cold ethanol, recrystallized from dioxane and air-dried. Yield: 3.18 g (71 %).
Colorless crystals, m. p. 149-150 ºC (dec.) from dioxane.
1H NMR (300 MHz, d6-DMSO):
2.23 (s, 3H, PhCH3), 2.43 (d, 1H, J=17.0 Hz, HCH),
2.96 (d, 1H, J=17.0 Hz, HCH), 3.44 (s, 3H, OCH3),
3.52 (d, 1H, J=12.2 Hz, H-3), 3.56 (s, 3H, OCH3),
3.60 (s, 3H, OCH3), 3.66 (s, 3H, OCH3),
3.94 (t, J1=J2=12.2 Hz, H-4), 4.38 (d, 1H, J=12.2 Hz, H-5),
4.67 (s, 1H, H-1), 5.40 (s, 1H, OH),
7.06 (d, 2H, J=8.0 Hz, 2' and 6'H arom.),
7.19 (d, 2H, J=8.0 Hz, 3' and 5'H arom.).
13C NMR (75 MHz, d6-DMSO):
41.4, 42.8, 51.1, 51.5, 51.6, 51.7, 54.3, 61.2, 62.8, 66.3, 74.3,
128.1 (2xC), 128.9 (2xC), 136.2,
136.6 (2xC), 167.7, 168.2, 169.5, 169.8.
FT IR (KBr, cm-1): 3511, 2953, 1729, 1516, 1495, 1364.
ESI MS [FIA in MeOH, CH3COONH4/CH3COOK]: 468.2 [M+NH4]+, 489.2 [M+K]+.
Reference
1. Haensel, W.; Haller, R. Arch. Pharm. (Weinheim Ger.) 1970, 303, 334-338.
.
 Dedicated to all moms in the world
Dedicated to all moms in the world


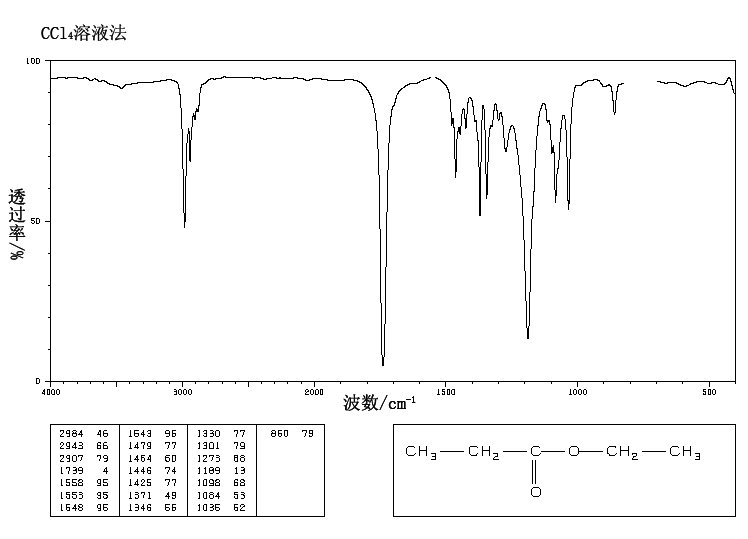
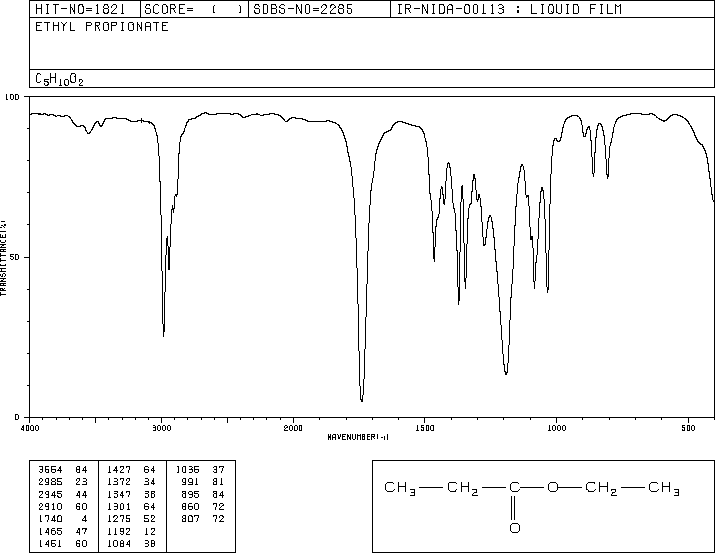


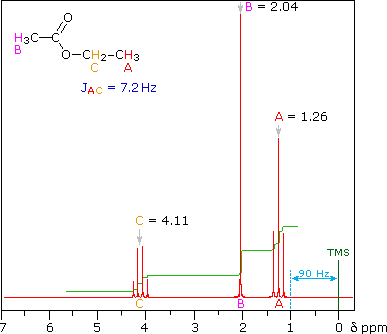
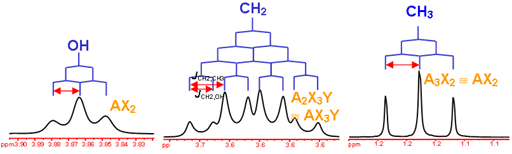
 This is the structure. See if you can assign the peaks on your own.
This is the structure. See if you can assign the peaks on your own. C has a higher chemical shift than D because it's closer to a more electron-withdrawing functional group.
C has a higher chemical shift than D because it's closer to a more electron-withdrawing functional group.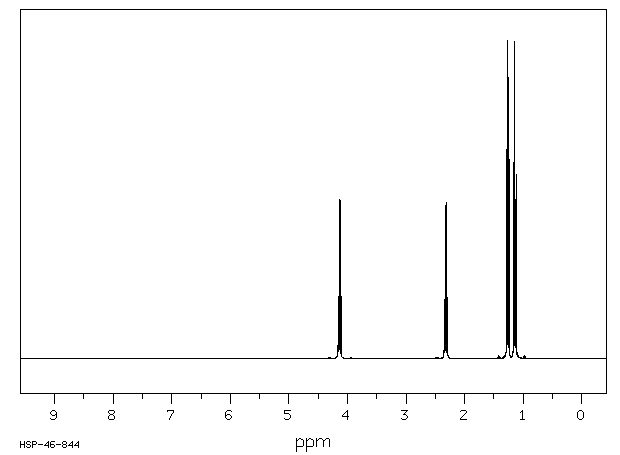

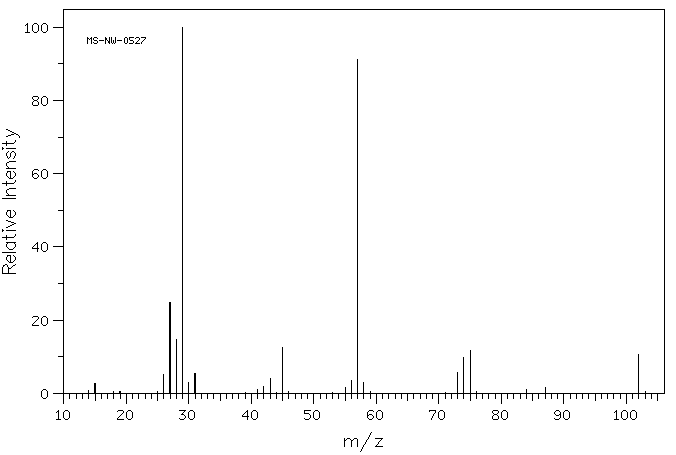
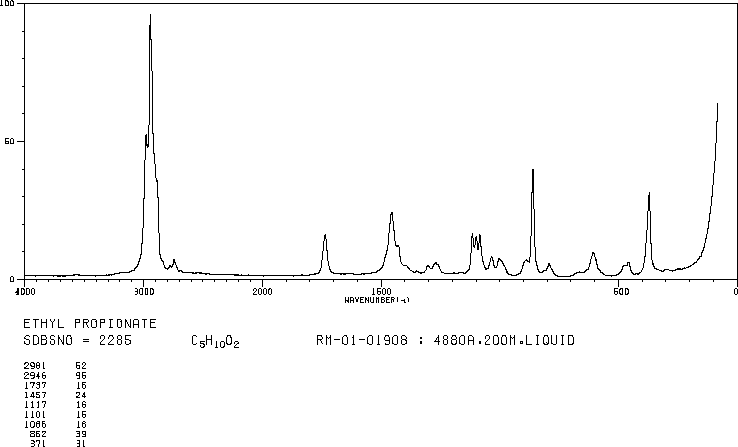


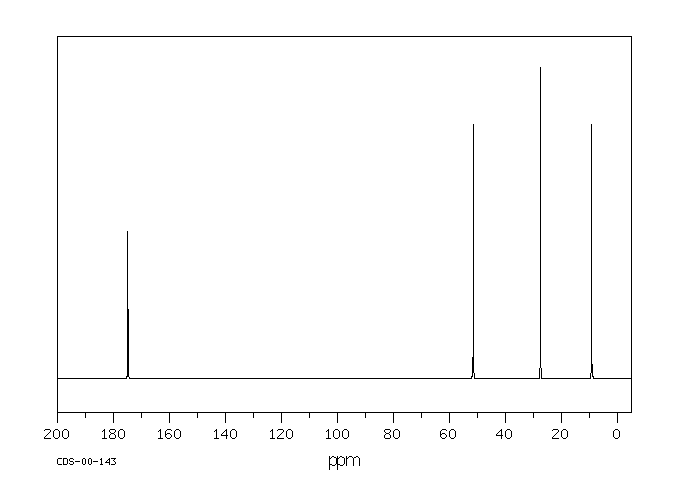



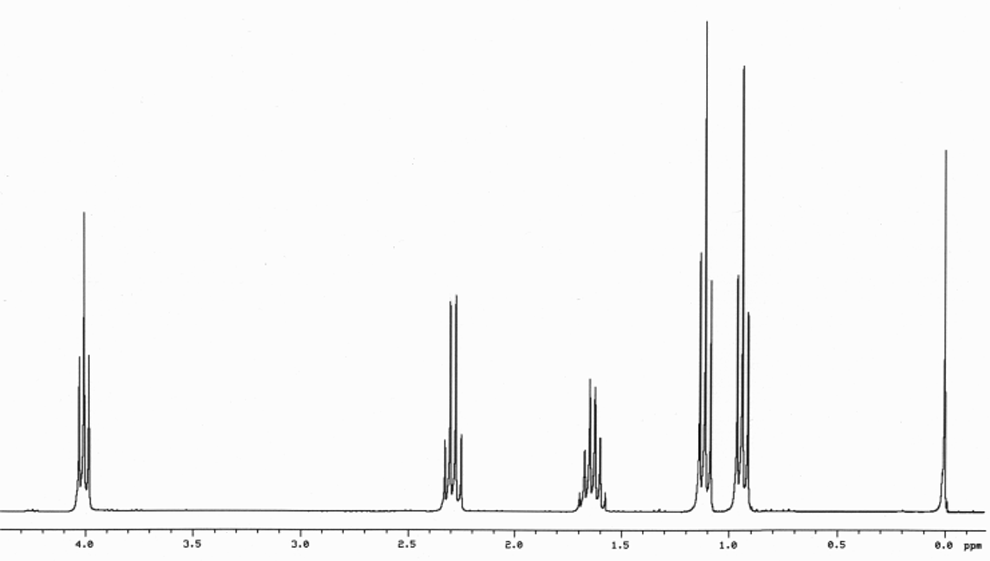






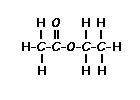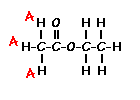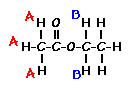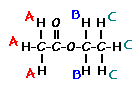
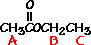




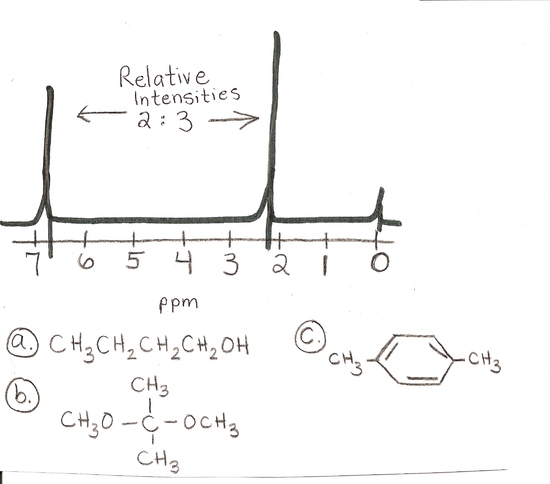
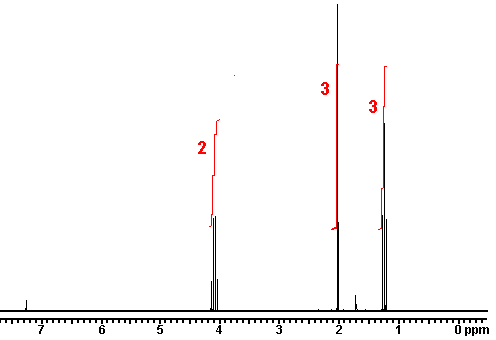
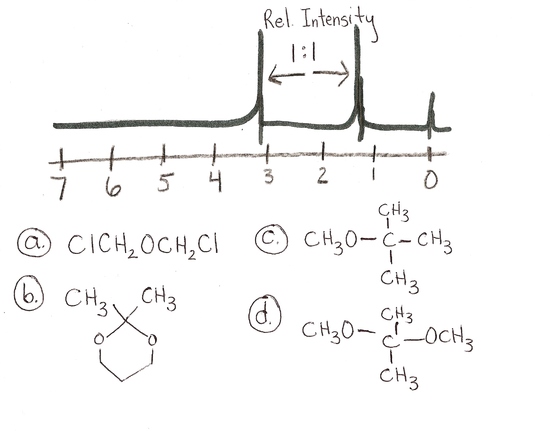 answer is a and d
answer is a and d