Organic Chemists from Industry and academics to Interact on Spectroscopy Techniques for Organic Compounds ie NMR, MASS, IR, UV Etc. Starters, Learners, advanced, all alike, contains content which is basic or advanced, by Dr Anthony Melvin Crasto, Worlddrugtracker, email me ........... amcrasto@gmail.com, call +91 9323115463 India skype amcrasto64
................DR ANTHONY MELVIN CRASTO Ph.D ( ICT, Mumbai) , INDIA 25Yrs Exp. in the feld of Organic Chemistry,Working for GLENMARK GENERICS at Navi Mumbai, INDIA. Serving chemists around the world. Helping them with websites on Chemistry.Million hits on google, world acclamation from industry, academia, drug authorities for websites, blogs and educational contribution
Pages
- Home
- ABOUT ME
- DIMENSIONS IN NMR SPECTROSCOPY
- 13 C NMR
- 1H NMR
- CHEMDOODLE/INTERACTIVE SPECT PREDICT
- Animations
- HELP ME
- Multinuclear NMR Spectroscopy
- Examples of 13C NMR
- Books on NMR spectroscopy
- UV-Visible Spectroscopy
- IR SPECTRA EXAMPLES
- Journals
- Organic spectroscopy site
- Spectroscopy sites
- IR SPECTROSCOPY
- Books-2
- Recommended Web Sites for Spectra and Spectrum-rel...
- DISCLAIMER
- Mössbauer spectroscopy
- FINDING CHEMICAL SPECTRA
- Mass Spectrometry
- NMR Overview
- Characterisation of Organic Compounds
- SDBS Spectral Database System for Organic Compounds
- CHEMICAL SHIFT
- MASS SPECTROSCOPY
- Books-1
- MASSBANK PORTAL
- 11B NMR
Wednesday 2 November 2016
Sunday 30 October 2016
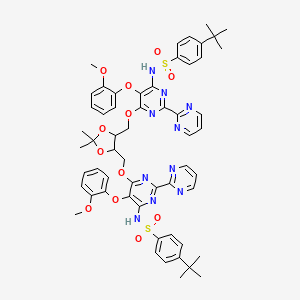
BOSENTAN PRECURSOR
| Molecular Formula: | C57H60N10O12S2 |
|---|---|
| Molecular Weight: | 1141.285 g/mol |
4-tert-butyl-N-[6-[[5-[[6-[(4-tert-butylphenyl)sulfonylamino]-5-(2-methoxyphenoxy)-2-pyrimidin-2-ylpyrimidin-4-yl]oxymethyl]-2,2-dimethyl-1,3-dioxolan-4-yl]methoxy]-5-(2-methoxyphenoxy)-2-pyrimidin-2-ylpyrimidin-4-yl]benzenesulfonamide
N,N′-(6,6′-(2,2-Dimethyl-1,3-dioxolane-4,5-diyl)bis-
(methylene)bis(oxy)bis(5-(2-methoxy phenoxy)-2,2′-bipyrimidine-6,4-diyl))bis(4-tert-butylbenzenesulfonamide)
Mp: 72−74 °C.
1
H NMR (400
MHz, CDCl3): δ 1.25 (6H, s), 1.29 (18H, s), 3.84−3.90 (4H,
m), 4.27−4.31 (2H, m), 6.84−6.87 (3H, t), 6.97−7.00 (2H,
dd), 7.09−7.13 (3H, t), 7.43−7.45 (10H, m), 9.0−9.01 (4H,
d), 8.43 (2H, br s);
13C NMR (100 MHz, CDCl3): δ 25.88,
30.02, 34.10, 55.01, 61.53, 77.36, 108.43, 111.4, 118.73, 120.4,
124.09, 124.34, 126.67, 127.38, 128.35, 135.30, 138.25, 144.74,
148.62, 150.99, 156.07, 156.71, 160.56;
MS: m/z 1142.2 (M +
H);
Elem. Anal: Found: C 59.87, H 5.20, N 12.38; Calcd for
C57H60N10O12S2: C 59.99, H 5.30, N 12.27
A new and efficient synthetic process for the synthesis of an endothelin receptor antagonist, bosentan monohydrate, involves the coupling of p-tert-butyl-N-(6-chloro-5-(2-methoxy phenoxy)-2,2′-bipyrimidin-4-yl)benzenesulfonamide (7) with (2,2-dimethyl-1,3-dioxolane-4,5-diyl)dimethanol (14) as a key step. This new process provides desired bosentan monohydrate (1) with better quality and yields. Our new methodology consists of technical innovations/improvements which totally eliminate the probability for the formation of critical impurities such as pyrimidinone 8, dimer impurity 9, and N-alkylated impurity 13 in the final drug substance.
Org. Process Res. Dev., 2013, 17 (8), pp 1021–1026
DOI: 10.1021/op400100s
/////////////
CC1(OC(C(O1)COC2=NC(=NC(=C2OC3=CC=CC=C3OC)NS(=O)(=O)C4=CC=C(C=C4)C(C)(C)C)C5=NC=CC=N5)COC6=NC(=NC(=C6OC7=CC=CC=C7OC)NS(=O)(=O)C8=CC=C(C=C8)C(C)(C)C)C9=NC=CC=N9)C
CC1(OC(C(O1)COC2=NC(=NC(=C2OC3=CC=CC=C3OC)NS(=O)(=O)C4=CC=C(C=C4)C(C)(C)C)C5=NC=CC=N5)COC6=NC(=NC(=C6OC7=CC=CC=C7OC)NS(=O)(=O)C8=CC=C(C=C8)C(C)(C)C)C9=NC=CC=N9)C
Heck–Matsuda Reaction in Flow
Product 3 was obtained as a mixture of diastereomers (58:42). The NMR data are consistent with literature precedent.20a
Major diastereomer: 1H NMR (300 MHz, CDCl3) δ (ppm) 7.25-7.28 (m, 2H), 7.14-7.17 (m, 2H), 5.14 (dd, 1H, J = 2.5, 5.8 Hz), 4.29 (t, 1H, J = 8.3 Hz), 3.79 (dd, 1H, J = 6.9, 8.4 Hz), 3.54-3.62 (m, 1H), 3.38 (s, 3H), 2.32 (dd, 1H, J = 7.7, 12.9 Hz), 2.04 (ddd, 1H, J = 5.1, 9.3, 13.1 Hz);
Minor diastereomer: 1H NMR (300 MHz, CDCl3) δ 7.25-7.28 (m, 4H), 5.16 (d, 1H, J = 4.4 Hz), 4.17 (t, 1H, J = 8.1 Hz), 3.72 (dd, 1H, J = 8.5, 9.7 Hz), 3.42 (s, 3H), 3.32-3.36 (m, 1H), 2.59 (ddd, 1H, J = 5.5, 10.3, 13.7 Hz), 1.91 (ddd, 1H, J = 2.4, 7.7, 10.2 Hz);
13C NMR (75 MHz, CDCl3) δ (ppm) 141.4, 140.0, 132.4, 132.3, 129.1, 128.7, 128.7, 128.5, 105.7, 105.4, 73.7, 73.0, 54.9, 54.7, 43.6, 42.1, 41.4, 41.1.
(20) (a) Oliveira, C. C.; Angnes, R. A.; Correia, C. R. D. J. Org. Chem. 2013, 78, 4373. (b) Oliveira, C. C.; Pfaltz, A.; Correia, C. R. D. Angew. Chem. Int. Ed. 2015, 54, 14036.
The optimization of a palladium-catalyzed Heck–Matsuda reaction using an optimization algorithm is presented. We modified and implemented the Nelder–Mead method in order to perform constrained optimizations in a multidimensional space. We illustrated the power of our modified algorithm through the optimization of a multivariable reaction involving the arylation of a deactivated olefin with an arenediazonium salt. The great flexibility of our optimization method allows to fine-tune experimental conditions according to three different objective functions: maximum yield, highest throughput, and lowest production cost. The beneficial properties of flow reactors associated with the power of intelligent algorithms for the fine-tuning of experimental parameters allowed the reaction to proceed in astonishingly simple conditions unable to promote the coupling through traditional batch chemistry.
13C NMR PREDICT
/////////
2-chloro-N-(2-phenylethyl)acetamide
2-chloro-N-(2-phenylethyl)acetamide
2-Chloro-N-(2-phenylethyl)acet
- Molecular FormulaC10H12ClNO
- Average mass197.661
Acetamide, 2-chloro
n-(2-phenylethyl) 2
[13156-95-1]
PAPER
HETEROCYCLES
An International Journal for Reviews and Communications in Heterocyclic Chemistry
Web Edition ISSN: 1881-0942
Published online: 11th October, 2016
Paper | Regular issue | Prepress
DOI: 10.3987/COM-16-13538
■ A Concise and Highly Efficient Synthesis of Praziquantel as an Anthelmintic Drug
Zhezhou Yang, Lin Zhang, Huirong Jiao, Rusheng Bao, Weiwei Xu, and Fuli Zhang*
*Shanghai Institute of Pharmaceutical Industry, China State Institute of Pharmaceutical Industry, 285 Gebaini Road, Shanghai 201203, China
Abstract
A concise and practical synthesis of praziquantel as anthelmintic drug is described. The key steps include a monoalkylation of ethanolamine for the preparation of 2-(2-hydroxyethylamino)-N-phenethylacetamide and a mild oxidation protocol with SO3-Py/DMSO as oxidant to transform alcohol into the corresponding aza-acetal. The telescoped synthesis is composed of five steps without purification of the intermediates, providing an overall yield of 80% with 99.8% purity after crystallization.
//////////////////
Saturday 29 October 2016
A Concise and Highly Efficient Synthesis of Praziquantel as an Anthelmintic Drug
PAPER
HETEROCYCLES
An International Journal for Reviews and Communications in Heterocyclic Chemistry
Web Edition ISSN: 1881-0942
Web Edition ISSN: 1881-0942
Published online: 11th October, 2016
Paper | Regular issue | Prepress
DOI: 10.3987/COM-16-13538
■ A Concise and Highly Efficient Synthesis of Praziquantel as an Anthelmintic Drug
Zhezhou Yang, Lin Zhang, Huirong Jiao, Rusheng Bao, Weiwei Xu, and Fuli Zhang*
*Shanghai Institute of Pharmaceutical Industry, China State Institute of Pharmaceutical Industry, 285 Gebaini Road, Shanghai 201203, China
Abstract
A concise and practical synthesis of praziquantel as anthelmintic drug is described. The key steps include a monoalkylation of ethanolamine for the preparation of 2-(2-hydroxyethylamino)-N-phenethylacetamide and a mild oxidation protocol with SO3-Py/DMSO as oxidant to transform alcohol into the corresponding aza-acetal. The telescoped synthesis is composed of five steps without purification of the intermediates, providing an overall yield of 80% with 99.8% purity after crystallization.
///////////////
2-bromocyclohexane-1,3-dione, Mom will teach you NMR
Mom will teach you NMR,So easy
1H NMR (CDCl3, 300 MHz): δ 6.63(s, 1H), 2.61(t, J = 5.9 Hz, 4H), 2.03 (p, J = 5.9 2 Hz, 2H).
BLACK =1H NMR
RED = 13C NMR
1H NMR PREDICT
13C NMR PREDICT


 COCK SAYS MOM CAN TEACH YOU NMR
COCK SAYS MOM CAN TEACH YOU NMR//////////////
Friday 28 October 2016
1-(biphenyl-4-yl)ethanone
Simultaneous rapid reaction workup and catalyst recovery
Green Chem., 2016, 18,5769-5772
DOI: 10.1039/C6GC02448C, Communication
DOI: 10.1039/C6GC02448C, Communication
Zhichao Lu, Zofia Hetman, Gerald B. Hammond, Bo Xu
By combining reaction work-up and catalyst recovery into a simple filtration procedure we have developed a substantially faster technique for organic synthesis.
By combining reaction work-up and catalyst recovery into a simple filtration procedure we have developed a substantially faster technique for organic synthesis.
By combining reaction work-up and catalyst recovery into a simple filtration procedure we have developed a substantially faster technique for organic synthesis. Our protocol eliminates the time-consuming conventional liquid–liquid extraction and is capable of parallelization and automation. Additionally, it requires only minimal amounts of solvent.
Simultaneous rapid reaction workup and catalyst recovery
*
Corresponding authors
a
Department of Chemistry, University of Louisville, Louisville, USA
E-mail: gb.hammond@louisville.edu
E-mail: gb.hammond@louisville.edu
b
College of Chemistry, Chemical Engineering and Biotechnology, Donghua University, 2999 North Renmin Lu, Shanghai 201620, China
E-mail: bo.xu@dhu.edu.cn
E-mail: bo.xu@dhu.edu.cn
Green Chem., 2016,18, 5769-5772
DOI: 10.1039/C6GC02448C
http://pubs.rsc.org/en/Content/ArticleLanding/2016/GC/C6GC02448C?utm_source=feedburner&utm_medium=feed&utm_campaign=Feed%3A+rss%2FGC+%28RSC+-+Green+Chem.+latest+articles%29#!divAbstract
. General procedure for a reaction Step 1. Reaction setup. The reaction is conducted in the usual way with the supported catalyst. Porelite® (typically 1 mL for every 0.1 gram of product) is added to the reaction mixture under stirring, Step 2. Reaction quench and rigid solvent extraction. If needed, the reaction is quenched with a suitable aqueous solution (e.g. NaHCO3 solution). • If the solvent used in the reaction is water-miscible (eg., DMF, methanol, etc.), a minimum amount of water immiscible solvent (e.g. 3 mL ether for every 1 g of product) is added to help organic material become entrenched in Porelite. • If the reaction is conducted in a water immiscible solvent (e.g. toluene, DCM), no extra solvent is needed in most cases. The excess amount of solvent is removed by rotavapor or by nitrogen/air purging (no need to remove the water from the mixture). The reaction mixture is filtered to remove aqueous-soluble components (starting materials, by-products, etc.) and washed with water (or HCl or Na2CO3 solution to remove basic or acidic byproducts. Vacuum is applied to dry the filtrate for 2 minutes to remove any remaining aqueous and volatile solvents. (For automatic flash chromatographic separation, an empty loading cartridge can be used, which can be directly attached to the commercial system. For manual chromatographic separation, a regular Büchner filter can be used). Step 3. Sample loading to chromatographic system. • The loading cartridge can be directly attached to the commercial flash chromatographic system (e.g., CombiFlash Rf series). • For manual chromatographic separation, the polymer powder is loaded directly onto a manual flash silica gel column (dry loading).
Because the polymer pad may contain some trapped air, it is recommended to start with the least polar solvent (e.g., hexane) during chromatographic separation to remove the trapped air.
1-(biphenyl-4-yl)ethanone
1-(biphenyl-4-yl)ethanone
Han, W.; Liu, C.; Jin, Z.-L. Organic Letters 2007, 9, 4005-4007
////////
Subscribe to:
Posts (Atom)