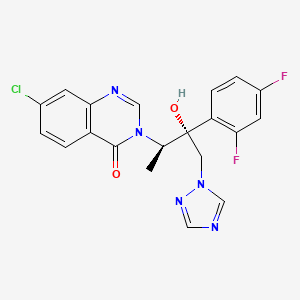
Methyl 2-(but-3-en-1-yl)oxazole-4-carboxylate
: Rf 0.39 (80% EtOAc in PE);
IR (neat, cm-1): 2955, 1733, 1642, 1586, 1437, 1322, 1196, 1142, 1109, 1001, 917, 805, 768;
1H NMR (500 MHz, CDCl3):
δ = 8.13 (1H, s; H3),
5.81 (1H, tdd, J = 6.5, 10.1, 17.0; H7),
5.05 (1H, qd, J = 1.4, 17.0; H8a),
4.99 (1H, qd, J =1.4, 10.1; H8b),
3.90 (3H, s; H9), 2.90 (2H, t, J = 7.8; H5),
2.52 (2H, tdd, J = 1.4, 6.5, 7.8; H6);
13C NMR (125 MHz, CDCl3):
δ = 165.2 (C4), 161.7 (C1), 143.7 (C3), 136.0 (C7), 133.1 (C2), 116.2 (C8), 52.1 (C9), 30.7 (C6),
27.5 (C5);
HRMS (ESI) calculated for C9H12NO3+ [M+H]+ 182.0772, found 182.0801.
...................................................................................
DR ANTHONY MELVIN CRASTO
 MY BLOGS ON MED CHEM
MY BLOGS ON MED CHEM


 amcrasto@gmail.com
amcrasto@gmail.com
...................................................................................
DR ANTHONY MELVIN CRASTO

New Drug Approvals ALL ABOUT DRUGS WORLD DRUG TRACKER MEDICINAL CHEMISTRY INTERNATIONALDRUG SYN INTERNATIONALSCALEUP OF DRUGS ALL FOR DRUGS ON WEBEUREKAMOMENTS
.......................................................................
DR ANTHONY MELVIN CRASTO Ph.D
MOBILE-+91 9323115463
GLENMARK SCIENTIST , INDIA
web link
web link
ABOUTME, BRAND ANTHONYCRASTO, COLLECTION OF SITE LINKS, BRAVESITES,GRAVATAR, JIMDO, SKILLPAGES, MIXXT, APNACIRCLE, ZIC ZAC, ZING ME, SCOOP IT, scribd, Stumbleupon, Delicious, pinnterest, tumblr, Newsvine, SLIDESHARE, ACADEMIA.EDU, GOOGLE PLUS, FACEBOOK, TWITTER, ISSUU, DIIGO, YOLASITE, Authorstream, Symbaloo, ISSUU, BLOGLOVIN,FRIENDFEED, NEWSVINE
Congratulations! Your presentation titled “Anthony Cra sto Glenmark scientist, helping millions with websites” has just crossed MILLION views.
アンソニー 安东尼 Энтони 안토니 أنتوني
blogs are 

SCALEUP OF DRUGS, ALL FOR DRUGS ON WEB,
MY CHINA, VIETNAM AND JAPAN BLOGS
ICELAND, RUSSIA, ARAB
GROUPS
you can post articles and will be administered by me on the google group which is very popular across the world
OPD GROUPSPACES, SCOOP OCI, organic-process-development GOOGLE, TVINX, MENDELEY WDT,SCIPEOPLE OPD, EPERNICUS OPD, SYNTHETIC ORGANIC CHEMISTRYLinkedIn group, DIIGO OPD,LINKEDIN OPD, WDT LINKEDIN, WDTI ZING